Ammonium Chloride Corrosion in Hydrotreating Units
Description of Damage
Ammonium chloride corrosion is characterized by general or localized corrosion, often pitting, normally occurring under ammonium chloride or amine salt deposits, often in the absence of a free water phase. The salts have a whitish, greenish or brownish appearance. Corrosion due to the deposition of ammonium chloride salts is only a problem when the salts can hydrate. Ammonium chloride is very hygroscopic. The salts can hydrate and become very corrosive to carbon steel, even at a process temperature above the dew point, at relative humidity as low as 20%. The temperature difference between 20% humidity and water dewpoint can be 30-50 oC, representing a significant pipe length with potential for severe corrosion. At temperatures below the dewpoint, the deposits will be washed away by the condensed water.
The attached references from Refincor summarize descriptions, locations of damage, corrosion rates and rules of thumb for corrosion by ammonium chloride salts reported by other refiners in more than 50 years of documenting NACE T-8 Refining Committee open discussions on corrosion in refineries.
Our online ammonium deposition calculations are a useful tool to evaluate the likelihood of ammonium bisulfide corrosion and the need for wash water injection.
Affected Materials
All commonly used materials are susceptible, in order of increasing resistance: carbon steel, low alloy steels, 300 series SS, Alloys 400, duplex SS, 800, and 825, Alloys 625 and C276 and titanium. The 300 series stainless steels can also experience chloride stress corrosion cracking in contact with ammonium chloride deposits.
Environmental Factors
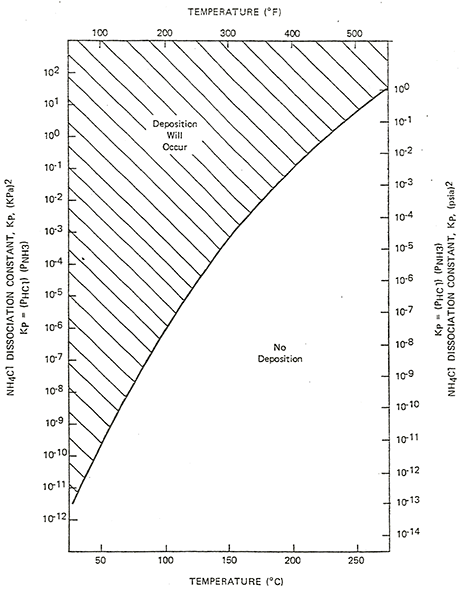
- Concentration (NH3, HCl, H20 or amine salts), temperature and water availability are the critical factors.
- Ammonium chloride salts may precipitate from high temperature streams as they are cooled, depending upon the concentration of NH3 and HCl, and may corrode piping and equipment at temperatures well above the water dewpoint [~300oF (149oC)]. The curve shows where deposition of ammonium chloride salts are predicted, based on the partial pressures of ammonia and hydrogen chloride.
- Ammonium chloride salts are hygroscopic, and readily absorb water. A small amount of water can lead to very aggressive corrosion ~360 mpy, depending on the temperature.
- Ammonium chloride and amine hydrochloride salts are highly water soluble, highly corrosive and form an acidic solution when mixed with water. Some neutralizing amines react with chlorides to form amine hydrochlorides that can act in a similar fashion.
Affected Equipment
Reactor effluent streams are subject to ammonium chloride salt fouling and corrosion at temperatures up to ~300F. Corrosion due to ammonium chloride salts is more difficult to predict than with ammonium bisulfide salts due to the higher temperature limits and unpredictability of deposition location, based on temperature and HCL/ammonia partial pressures. Inspecting for ammonium chloride corrosion is best done based on prior inspection observations, process stream sampling, input from process engineers and industry experience. If the inlet feed has negligible nitrogen content, then ammonium chloride salt corrosion may not be active. The attached Refincor references document the experience of other refiners with proven and suspected ammonium chloride salt corrosion. Locations of detected ammonium chloride salt corrosion included HDS effluent exchangers and a hydrotreater desulfurizer prefractionator feed/bottoms exchanger.